
Nearly every clock you interact with — from the watch on your wrist to the systems powering GPS and the internet — operates on the same fundamental principle: tracking a regular, repeating motion.
A good clock needs a reliable tick. In a pendulum clock, that tick is the steady, rhythmic swing of the pendulum — back and forth, back and forth, at a near-constant rate.
Our team of physicists is investigating whether an even more precise clock could be built using the atomic nucleus. Today's most accurate timekeepers already exploit the quantum properties of atoms, but a clock rooted in the nucleus — the tiny, dense core at an atom's center — could theoretically maintain a steadier rhythm. That's because the nucleus is far less susceptible to environmental disturbances like temperature fluctuations or stray electromagnetic fields. In our research, published in Nature, we measured and interpreted a unique nuclear property of thorium-229 embedded in a crystal — a finding that could bring nuclear clocks meaningfully closer to reality.
Ultraprecise clocks are far more than laboratory curiosities. They underpin navigation systems, global communications infrastructure, and international timekeeping standards. And pushing the boundaries of timing accuracy routinely opens unexpected doors in fundamental science.
How atomic clocks work
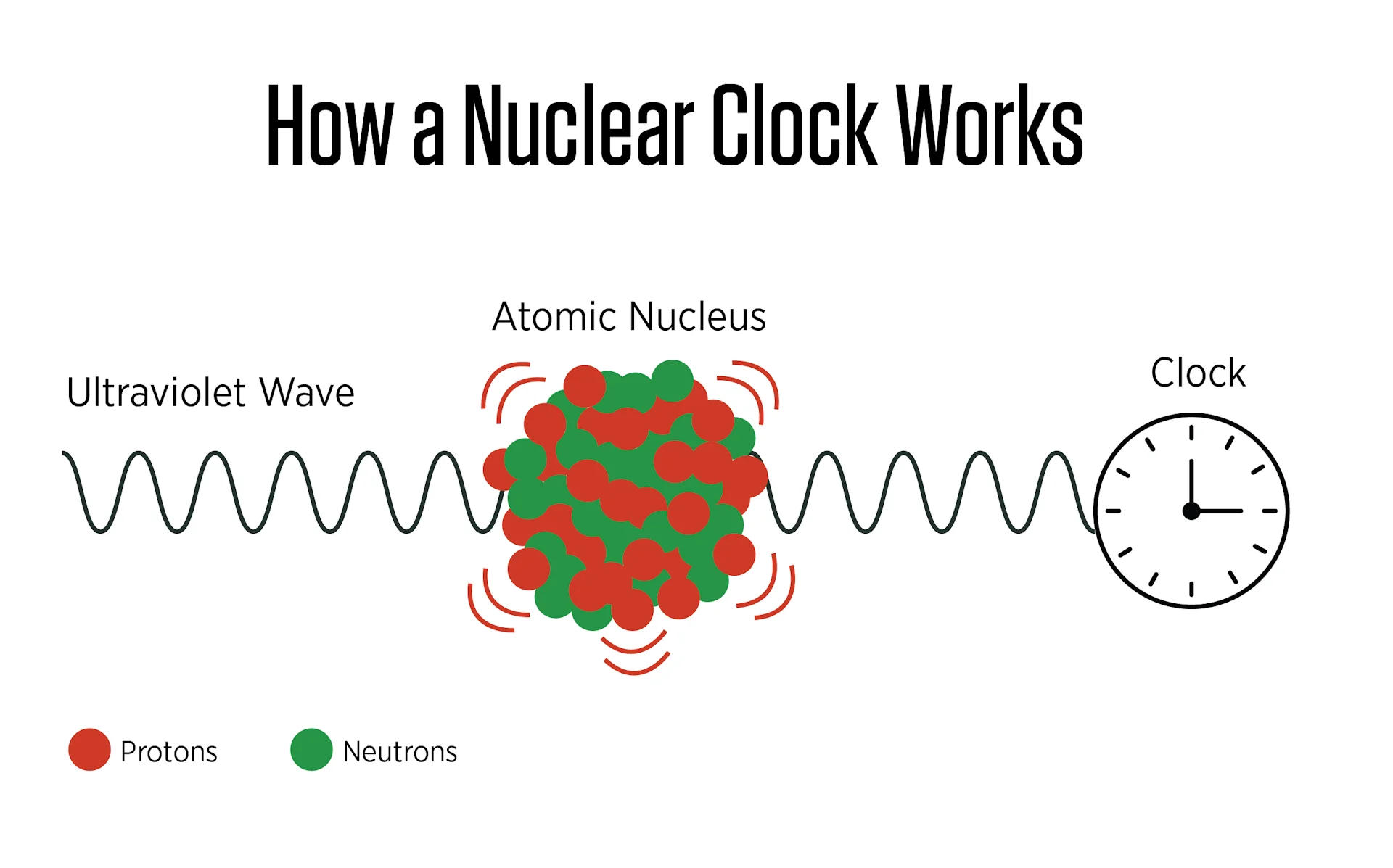
In an atomic clock, researchers shine a precisely tuned laser onto a material and adjust the light's frequency until it triggers a specific atomic response — typically by nudging an electron from one energy level to another. The moment this happens, the atoms absorb the laser light most strongly, providing a clear signal that the frequency is exactly right.
That absorption peak occurs at an extraordinarily precise frequency. Frequency describes how often something repeats in a given time. For a pendulum, it's the number of complete swings per second. For light, it's the number of wave cycles passing a fixed point each second — a value that also determines the light's energy and, within the visible spectrum, its color.
By detecting when atoms absorb laser light most strongly, scientists effectively use the laser as a metronome — counting light waves rather than pendulum swings.
Keeping that tick rate constant requires precisely matching the laser's energy to the electron excitation energy of the chosen atom. Because those energy levels are fixed by the laws of physics, atomic clocks built around the same atom will tick at identical rates anywhere in the universe — a universality that makes them uniquely powerful.
But atomic clocks aren't immune to error. Any environmental factor that shifts an atom's energy levels — an unaccounted-for magnetic field, a change in room temperature — will cause the clock to tick at a subtly different rate.
That vulnerability points toward a more robust alternative. Deep within every atom lies something far smaller: the nucleus. Current atomic clocks track electron behavior on the atom's outer shell. A nuclear clock, by contrast, would use an energy transition within the nucleus itself. Because a nucleus is roughly 10,000 times smaller than the atom it inhabits, it is substantially less sensitive to temperature swings, electric fields, and other outside interference — making it an appealing foundation for a far more stable clock.
The challenge is that nature doesn't make such a clock easy to build. A unique property we identified in our research could change that.
What makes thorium-229 special?
In an exceptionally rare quirk of nuclear physics, thorium-229 exists in two closely spaced energy states: a ground state and a slightly elevated excited state. These represent two distinct configurations of the nucleus, and scientists can use lasers to drive the nucleus between them — exactly the kind of controllable transition needed to build a clock.
The first hurdle was determining precisely how much energy is required to push the thorium-229 nucleus into its excited state — a measurement that took nearly 50 years to accomplish. Our group and others finally achieved this in 2024, pinning down a transition frequency of roughly 2 quadrillion — 2 × 1015 — cycles per second.
The next challenge: confirming that the nucleus has actually been excited. Until recently, physicists assumed the best approach was to watch for the faint flashes of light that excited nuclei typically release. But two significant obstacles stand in the way.
First, in most materials, thorium nuclei don't release their energy as light at all. Instead, through a process called internal conversion, the energy is transferred directly to a nearby electron in the surrounding material.
Second, even in cases where light is emitted, it falls in the vacuum ultraviolet range — a part of the electromagnetic spectrum that air readily absorbs and that is notoriously difficult to detect.

A different way to 'listen' to the nucleus
In our work, we inverted the problem entirely. Rather than chasing the light the nucleus emits, we looked directly for the internal conversion electrons it produces.
We deposited an ultrathin layer of thorium dioxide — just a few dozen atoms thick — onto a small metal disc. A laser tuned to the resonant energy excited the thorium nuclei in the sample. As those nuclei relaxed, they transferred their energy to nearby electrons, some of which escaped the surface. Using carefully arranged electric and magnetic fields, we guided those liberated electrons into a detector.
By sweeping the laser across a range of frequencies and counting the detected electrons at each step, we could map how closely the laser energy matched the nuclear excitation energy. When the match was exact, the signal emerged clearly in the data, revealing the precise frequency at which thorium-229 nuclei absorb most strongly.
We also measured how long the excited nuclear state persisted in this material before decaying — providing a direct window into how the surrounding solid influences the nucleus's behavior.
The experimental results become substantially more powerful when combined with theoretical calculations. Computational models can estimate how the host material shifts the energy required to excite thorium and how efficiently the material converts nuclear energy into emitted electrons. These calculations allow researchers to disentangle the nucleus's intrinsic properties from effects imposed by its solid-state environment — a distinction that is essential for designing practical nuclear clocks.
Why this approach matters
Detecting electrons instead of photons offers two concrete advantages.
First, it dramatically expands the range of materials suitable for hosting a nuclear clock. Earlier techniques worked best in materials where electrons were difficult to dislodge, severely limiting researchers' options. Our method lifts that constraint, opening up a much broader palette of materials that were previously impractical — and potentially easier to incorporate into compact clock designs.
Second, this approach could enable a fundamentally simpler style of nuclear clock. Rather than relying on sensitive optical detectors, a clock built on this principle could read out time by measuring a tiny electrical current generated by the emitted electrons — a far more hardware-friendly output that could make miniaturization significantly more tractable.
What could nuclear clocks be used for?
Beyond improving everyday timekeeping, nuclear clocks could one day give researchers a powerful tool for probing the deepest questions in physics — including whether the fundamental constants of nature are truly constant over cosmic timescales, or whether subtle signatures of new phenomena, such as dark matter, might be detectable through their effect on atomic transitions. More stable clocks would also benefit technologies that depend on tightly synchronized timing, including next-generation navigation and communications systems.
Our work represents an early but meaningful step in that direction. It does not deliver a finished clock — but it removes a practical experimental barrier and equips researchers with a new technique for studying how the thorium nucleus behaves inside solid-state materials.
Eric R. Hudson receives funding from ARO, DARPA, NIST, NSF, and RCSA.
Andrei Derevianko receives funding from NASA and National Science Foundation.


